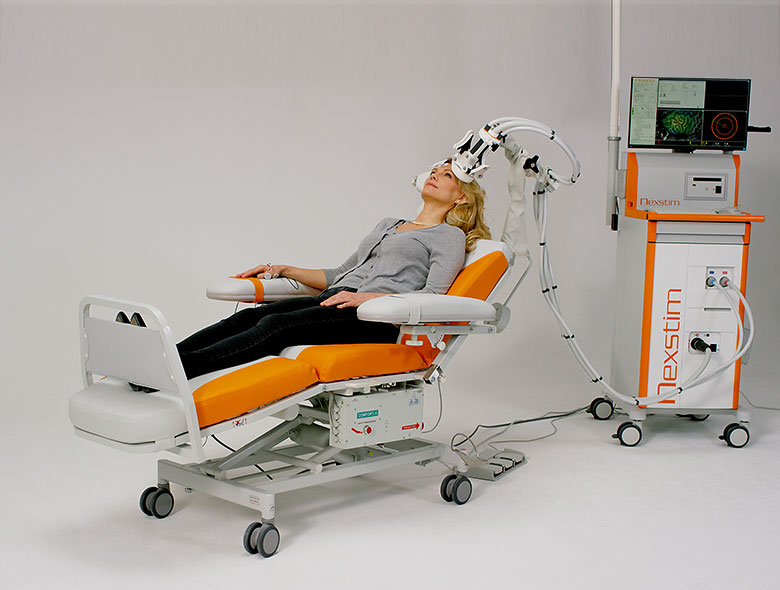
Indications for use in depression and pain
Depression
Nexstim's noninvasive NBT® System is cleared by the Food and Drug Administration (FDA, K171902). Nexstim Navigated Brain Therapy (NBT) System 2 is indicated for the treatment of Major Depressive Disorder in adult patients who have failed to achieve satisfactory improvement from prior antidepressant medication in the current episode.
CE mark: Nexstim Navigated Brain Therapy System for depression is intended to be used for treatment of major depressive disorder (MDD) by targeting and delivering noninvasive repetitive TMS stimulation to the patient's dorsolateral prefrontal cortex.
Pain
The Nexstim NBT System is not cleared by the FDA for commercial use of the treatment of chronic pain in the United States, for investigational use only.
CE mark Intended use
Nexstim Navigated Brain Therapy System is intended to be used for localization and assessment of the primary motor cortex for pre-procedural planning purposes. In adult patients suffering from chronic unilateral neuropathic pain, Nexstim NBT System is intended to provide electric field navigated noninvasive repetitive TMS stimulation as therapy to alleviate pain.
CE mark Indications for use
Nexstim MRI-Guided and E-Field Navigated Brain Therapy System (Nexstim NBT® System) is indicated for noninvasive mapping of the primary motor cortex of the brain to its cortical gyrus. Nexstim NBT System is indicated for MRI-guided and electric field (or E-field) navigated, noninvasive, repetitive TMS stimulation (rTMS) of the motor cortex as therapy to alleviate chronic unilateral neuropathic pain in adult patients.
Nexstim NBT® System is intended to be used by trained clinical professionals.